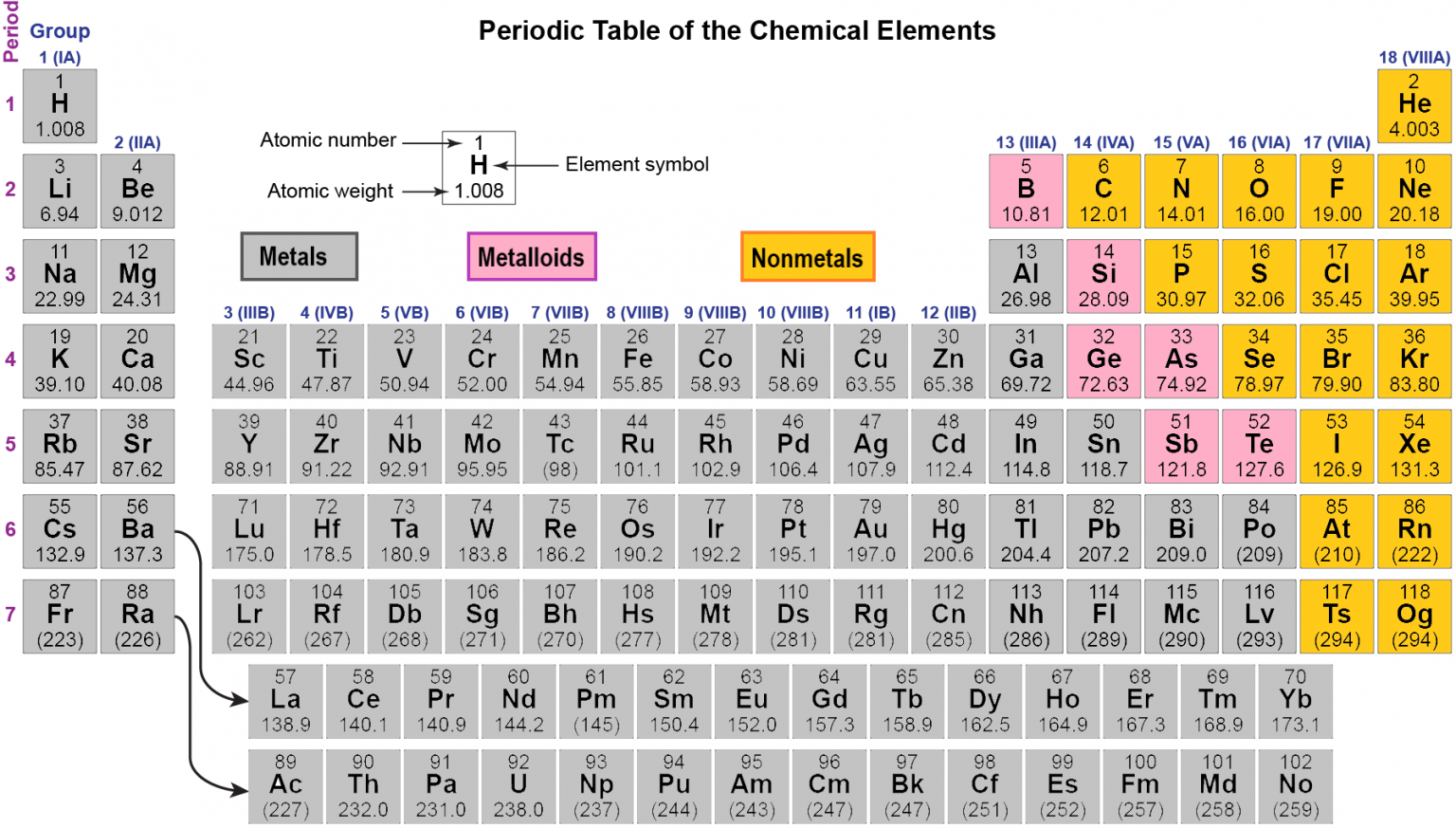
Many alternative representations of the periodic law exist, and there is some discussion as to whether there is an optimal form of the periodic table.Įach chemical element has a unique atomic number ( Z ) representing the number of protons in its nucleus. Some scientific discussion also continues regarding whether some elements are correctly positioned in today's table. It is not yet known how far the table will go beyond these seven rows and whether the patterns of the known part of the table will continue into this unknown region. 
Today, while all the first 118 elements are known, thereby completing the first seven rows of the table, chemical characterisation is still needed for the heaviest elements to confirm that their properties match their positions. In nature, only elements up to atomic number 94 exist to go further, it was necessary to synthesise new elements in the laboratory. The periodic table continues to evolve with the progress of science. The periodic table and law are now a central and indispensable part of modern chemistry. Seaborg's discovery that the actinides were in fact f-block rather than d-block elements. A recognisably modern form of the table was reached in 1945 with Glenn T. It was explained early in the 20th century, with the discovery of atomic numbers and associated pioneering work in quantum mechanics both ideas serving to illuminate the internal structure of the atom. The periodic law was recognized as a fundamental discovery in the late 19th century. As not all elements were then known, there were gaps in his periodic table, and Mendeleev successfully used the periodic law to predict some properties of some of the missing elements. The first periodic table to become generally accepted was that of the Russian chemist Dmitri Mendeleev in 1869 he formulated the periodic law as a dependence of chemical properties on atomic mass. Nonmetallic character increases going from the bottom left of the periodic table to the top right. Metallic character increases going down a group and decreases from left to right across a period. Vertical, horizontal and diagonal trends characterize the periodic table. Elements in the same group tend to show similar chemical characteristics. The table is divided into four roughly rectangular areas called blocks. It is a depiction of the periodic law, which says that when the elements are arranged in order of their atomic numbers an approximate recurrence of their properties is evident. It is an icon of chemistry and is widely used in physics and other sciences. Not to miss out, element 102, Nobelium, No, is named in honor of Alfred Nobel, who set aside his vast fortune to establish Nobel Prizes.The periodic table, also known as the periodic table of the elements, arranges the chemical elements into rows (" periods") and columns (" groups"). Mendeleev never received a Nobel Prize for his work, but element 101 was named Mendelevium, Md, after him. For instance they discovered phosphorus when they isolated it from urine. Over time these gaps have gradually been filled in as scientists unearthed new elements. Not only did Mendeleev arrange the elements in the correct way, but he also had the foresight to leave gaps for undiscovered elements. At that time, he had only 50 elements to arrange. He wrote the properties of the elements on pieces of card and rearranged them until he realised that, by putting them in order of increasing atomic weight, certain properties of elements regularly occurred. Then in 1869, a Russian scientist called Dmitri Mendeleev produced one of the first practical periodic tables. Several other attempts were made to group elements together over the coming decades. The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals and earths. The discovery of other elements followed regularly and soon it became necessary to arrange them in some sort of order. However, the first scientific discovery of an element occurred in 1649 when Hennig Brand discovered phosphorous. Copper has been used by humans for as much as 7000 years and elements such as gold, silver, tin, lead and mercury have been known for many thousands of years.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
